- Author: Konrad Mathesius
Summary Review
Results from this year's Italian ryegrass (IR) herbicide trials helped quantify differences in herbicide resistance among IR populations within the southern Sacramento Valley. The trial took place in Bird's Landing, CA (near Rio Vista), and was replicated farther north in Esparto, CA.
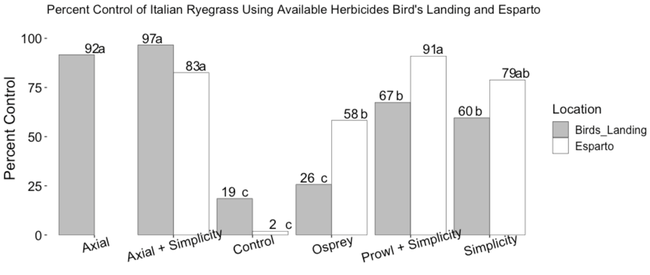
- Trials from this year suggest that Osprey-resistant IR populations often associated with the area around Dixon, CA could extend at least as far south as Bird's Landing with only 26% control of IR by Osprey in the trial site.
- The Osprey-resistant population appears to also be moderately resistant to Simplicity, another herbicide in the same chemical family, which only provided 60% control at the Bird's Landing site.
- Axial (an ACCase inhibitor) worked well at controlling IR in Bird's Landing (92% control).
- In Esparto, IR in the trial was somewhat more susceptible to ALS inhibitors. IR control was only around 80% with Simplicity.
Introduction
For a few years I've heard PCAs mention that Italian ryegrass (IR) populations around Dixon are showing resistance to Osprey (an ALS inhibitor herbicide), but quantification of differences in weed control can help provide a better understanding of what growers are dealing with. IR is notorious for its capacity to develop herbicide resistance to multiple modes of action (MoA). As an obligate outcrossing plant, IR must cross-pollinate in order to produce viable seed. This means that genetic material is regularly exchanged during pollination, which allows populations to respond to environmental pressure. Development of herbicide resistance in IR over the years has caused increasing concern for small grain growers, who are limited in their options for control.
This study is part of a wider range of studies examining different options available to growers for IR control in small grains. This study quantifies the efficacy of various available herbicides on IR populations in small grains both in the northern parts of Yolo County and the southern parts of Solano County.
Methods
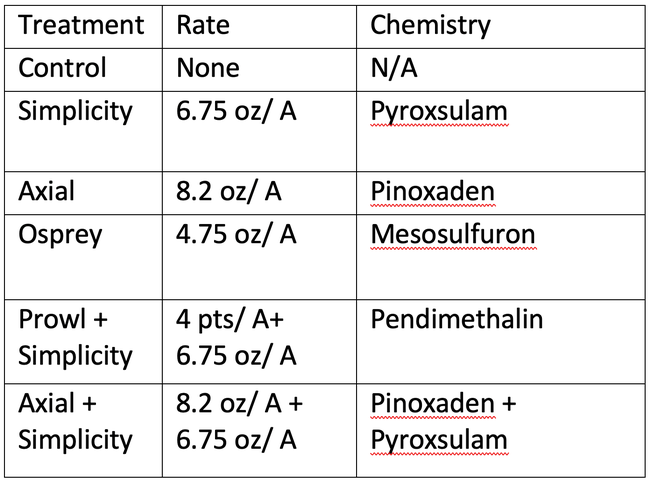
Two trials were set up: one in Bird's Landing and one in Esparto. Each plot was 15 x 100 feet and was replicated 4 times at each location. Plots were planted with wheat in late December 2022 after a burndown treatment and were treated in January of 2023 using the maximum label rate of each herbicide along with recommended adjuvants (Table 1). A 20ft x 15ft section of each plot was left unsprayed as an untreated reference. Axial was unintentionally applied at half label rate in Esparto.
Prowl was tested to see if it might improve control by by providing residual control of late germinating ryegrass that escaped preseason control efforts but did not germinate with the first fall rains. IR control was not significantly better in the Prowl tankmix vs Simplicity applied alone in this experiment.
Treatments and Rates Evaluated for Italian ryegrass Control in Two Wheat Sites in 2023 in the Sacramento Valley.
Weed counts were taken in late February in each plot (3 sub-samples). The untreated reference sections within each plot were measured once (1 representative sub-sample). Percent weed control was measured by the difference in ryegrass density within the treated and untreated areas in each plot.
Grain was hand harvested in late July using 3 x 4ft^2 quadrats in each plot. Spikes were collected by cutting the stem at the base of the spike using a sickle. Samples were then air dried. Grain yield was determined by subtracting an estimated chaff weight of 17% from the weight of the harvested spikes (McCartney et. al, 2006).
Results and Discussion
Percent control
Within each location, herbicides varied significantly in terms of the capacity to control IR.
In Bird's Landing (20 miles south of Dixon, CA), Axial provided significantly better IR control than Simplicity, Prowl + Simplicity, and Osprey. Osprey did not reduce IR populations compared to the non-treated control plots. (Figure 2)
In Esparto, a tank mix of Prowl + Simplicity provided better control of IR than Osprey but was not different than Simplicity alone (p = 0.52) or the tank mix of Axial + Simplicity (p = 0.81). Osprey provided only moderate control (58%) and was marginally different from Simplicity (p = 0.084), which provided 79% control.
The differences in control between the two ALS-inhibitor herbicides (Simplicity and Osprey) at the two locations are an indication of the variation among IR populations that are only 50 miles apart.
Yield
Yield results collected from the Bird's Landing site show trends that generally correspond with differences in weed control, although no significant differences in estimated yield were found due to the variability of the data.
Conclusion
Variations in herbicide efficacy are a good reminder that there is a lot of genetic variability in IR populations; even fields only a few miles apart could have important differences in response to herbicides. This means that grower practices can directly impact the development of herbicide resistance in their area. Growers in the Dixon area should incorporate IPM practices listed above and consider the use of Axial as an alternative weed control if they haven't already, and growers farther north should remain particularly vigilant about preserving the efficacy of their ALS inhibitor herbicides by integrating some of the IPM practices listed below.
- Using certified seed (to prevent weed seeds from hitching a ride to entirely different areas of the state)
- Thoroughly cleaning equipment, or operating equipment only within local areas to prevent the spread of weed seed to other parts of the state
- Rotating herbicides within the season and from one season to the next, where possible
- Spraying at the right time (check labels, apply on the early end of the spray window)
- Spraying the right rate (Axial applications in Esparto were sprayed unintentionally at half-rate and provided no control).
- Spraying when weeds are actively growing
- Planting wheat at the right density
- Incorporating mechanical cultivation or Harvest Weed Seed Control where possible
- Checking and calibrating spray nozzles
- Incorporate the use of pre-emergent herbicides labeled in California
- Rotating crops where possible to diversify herbicide programs
- Check for escapes and monitor fields for efficacy
Growers are also encouraged to take advantage of UC IPM resources online, and in-person through their local farm advisors.
References
McCartney, D.H.; Block, H.C.; Dubeski, P.L.; Ohama, A.J. Review: The composition and availability of straw and cha? from small grain cereals for beef cattle in western Canada. Can. J. Anim. Sci. 2006,86, 443–455.
- Author: Clebson G Goncalves Ph.D.
- Posted by: Gale Perez
In California, Italian ryegrass [Lolium perenne L. spp. multiflorum (Lam.) Husnot] has been around for a long time and is a major weed in orchards, vineyards, field crops, fallow fields, and so on. It grows vigorously in winter and early spring. Italian ryegrass is a short, rhizomatous, that grows from 11 to 35 inches tall, often with erect stems exhibiting purple coloration at the base. That species can be identified by its dark green, glossy, and hairless leaves that are rolled in the bud. Auricles are well-developed and the shape can vary from clasping to blunt, and the ligules are long and membranous. Once flowering occurs, Italian ryegrass is easily distinguishable by alternating spikelets that run along the length of the main seed head stem. The seed heads range from 3 to 12 inches and consist of solitary small stalkless spikelets (Fig. 1). Additional details regarding identification can be found on the UC IPM website.
Also called annual ryegrass, Italian ryegrass herbicide resistance and management strategies have been the topic of discussion among growers, PCAs, UC Cooperative Extension farm advisors, and UC weed science specialists. Because Italian ryegrass has been controlled in crop production systems mainly with herbicides for decades, since 1998, populations of Italian ryegrass have been documented to be resistant to herbicides (Fig. 2). In California, the first report of Italian ryegrass glyphosate-resistant was in 2008 in almond orchards and vineyards, and the evolution and spread of these populations in the state made alternative postemergence herbicides an important management strategy against this troublesome species (published by Jasieniuk et al. 2008, Weed Sci. 2008).
Repeated herbicide use has selected Italian ryegrass populations resistant to multiple of herbicide mode of actions. A recent Weed Science publication by Dr. Brad Hanson's lab confirmed multiple Italian ryegrass resistance to the herbicides paraquat (Gramoxone® 2.0 SL), clethodim (Envoy Plus®), and glyphosate (Roundup PowerMax®). A second study conducted in Dr. Hanson's lab also confirmed that another Italian ryegrass biotype population collected in Hamilton City, CA is resistant to paraquat (Gramoxone® 2.0 SL), clethodim (Envoy Plus®), glyphosate (Roundup PowerMax®), and Mesosulfuron-Methyl (Osprey®). It should be noted that ryegrass populations resistant to fluazifop-butyl (Fusilade® DX), glufosinate-ammonium (Rely® 280), and sethoxydim (Poast®) also have been reported in other cropping systems elsewhere in California.
Worldwide Italian ryegrass has been identified as resistant to 29 herbicides, from 8 different groups of classification with multiple resistance to 1, 2, 3, or 4 sites of action (WeedScience.org, 2022). The most recent Italian ryegrass biotype was identified in a soybean and winter wheat field in North Carolina with multiple resistance to 4 sites of action (clethodim, glyphosate, nicosulfuron, and paraquat).
Management Strategies
For efficient management of Italian ryegrass, integrated weed management (IWM) practices must be adopted that allow the combination of various control strategies, including preventive, cultural, mechanical, physical, biological, and chemical control.
Prevention Control: Avoiding dispersion and introduction of Italian ryegrass into the property and ensuring that seeds and equipment are free of Italian ryegrass contaminants is one of the main strategies in the IWM programs.
Physical Control: Soil tillage may be used as one strategy to be added to the IWM toolbox. Italian ryegrass seeds buried in the soil die quickly and must be close to the surface to emerge. So, this practice can help suppress the population and provide long-term control.
Chemical Control: Italian ryegrass control using PRE and POST herbicide management programs continues to be the most effective. A well-designed herbicide application program with a combination of PRE- and POST-emergent herbicides (e.g., fluazifop-butyl (Fusilade® DX), glufosinate-ammonium (Rely® 280), pyroxsulam (SimplicityMT), rimsulfuron (Matrix®SG), sethoxydim (Poast®), flumioxazin (Chateau®), oxyfluorfen (GoalTender®), pendimethalin (Prowl H2O), and so on are possible options) still been quite effective in controlling Italian ryegrass. Growers should always consult a PCA and a local UCCE farm advisor to help design herbicide application programs and application rates appropriate for their unique growing system. Growers should also keep in mind that they need to avoid repeated herbicide use to prevent resistance in Italian ryegrass populations. Herbicides with different modes of action on a rotational basis should be encouraged to suppress the Italian ryegrass population and provide long-term control.
The growers also may adopt non-synthetic herbicide products for Italian ryegrass control. Among those available commercial products, the active ingredients, including acetic acid (Weed Pharm®), citric acid + Clove oil (BurnOut®), caprylic acid + capric acid (Suppress®), pelargonic acid + related fatty acids (Scythe®), D-limonene (AvengerAG®), ammonium nonanoate (Axxe®) and others, have been used for the non-selective control of that weed. Usually, organic herbicides are recommended for grass and broadleaf weeds in the early stages of growth. For Italian ryegrass already established, organic herbicides have shown poor effectiveness, and recovery approximately two weeks after treatment application (Fig. 3). Studies have shown that organic herbicides applied to control Italian ryegrass may require late sequential application due to the non-systemic characteristics of these products (ongoing trials by Clebson Gonçalves, 2023).
Even with the increase in herbicide-resistant weeds, herbicide control options continue to be the most effective and used option for producers because of time and money. However, diversified management practices should be encouraged as they are crucial for crop productivity, efficiency, saving cost, and sustainability of the systems.
Clebson Gonçalves is the UC Cooperative Extension Diversified Agriculture Advisor in Lake and Mendocino counties.
- Author: Bradley Hanson
Sharing a link to an upcoming seminar on a broad screening of annual bluegrass (Poaannua) resistance to herbicides. This is related to aUSDA Specialty Crop Research Initiative grant called "ResistPoa".
From Weed Science Society of America press release:
Annual bluegrass (Poa annua) is a common and troublesome weed in turfgrass and other landscapes. Its invasive biology and acquired herbicide resistance challenges turfgrass managers by increasing costs and compromising expected outcomes. The USDA-SCRI (Specialty Crops Research Initiative) funded ResistPoa project (www.resistpoa.org) seeks to characterize the national distribution of herbicide-resistant P. annua and limit its impact as a weed on managed turfgrass systems through a coordinated research and outreach effort on its biology, resistance mechanisms, socioeconomic aspects, and alternative control methods.
Panelists will discuss the project, its findings, and the potential for future research. Chief among the findings of the project has been the discovery of almost overwhelming presence of resistant populations in certain regions and scenarios. More than 560 of 1,349 total populations screened were suspected resistant to at least one herbicide mode of action. Investigations also revealed socio-economic constraints that affect control strategies and stewardship of herbicide technologies. Panelists will discuss integrated management practices that were developed as part of this multi-state project that can be implemented for improved control and herbicide resistance mitigation.
X
20230523 OPMP Webinar - ResistPoa FINAL
- Author: Whitney Brim-DeForest
- Author: Taiyu Guan
- Author: Troy Clark
- Posted by: Gale Perez
From the CAPCA Adviser magazine :: June 2022
Whitney Brim-DeForest is a UCCE Rice and Wild Rice Advisor for Sutter, Yuba, Sacramento, and Placer Counties; Taiyu Guan is an Assistant Specialist in UCCE Sutter-Yuba Counties; Troy Clark is a Rice Junior Specialist for UCCE Butte County.
Introduction
In California rice, herbicide resistance has been documented in Echinochloa spp. since the early 2000's. Recent reports of uncontrolled grasses, as well as possible new species or biotypes have precipitated renewed research on this genus. Sixty-four watergrass samples were collected from a survey conducted in 2020, with grower and PCA-submitted samples from across the Sacramento Valley, as well as samples collected from University of California and Rice Experiment Station fields. Those samples were representative of all the watergrass species/biotypes: late watergrass, junglerice, barnyardgrass, and the new biotype/species. This experiment was a follow-up to our 2018 screening of watergrass (CAPCA Adviser, 2021).
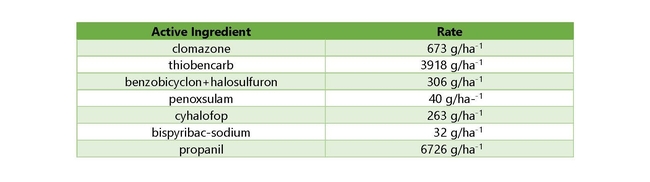
The overall objective of this study was to determine the distribution and status of resistance to currently-registered herbicides in these species (cyhalofop, propanil, bispyribac-sodium, penoxsulam, benzobicyclon+halosulfuron, clomazone, and thiobencarb).
Methods
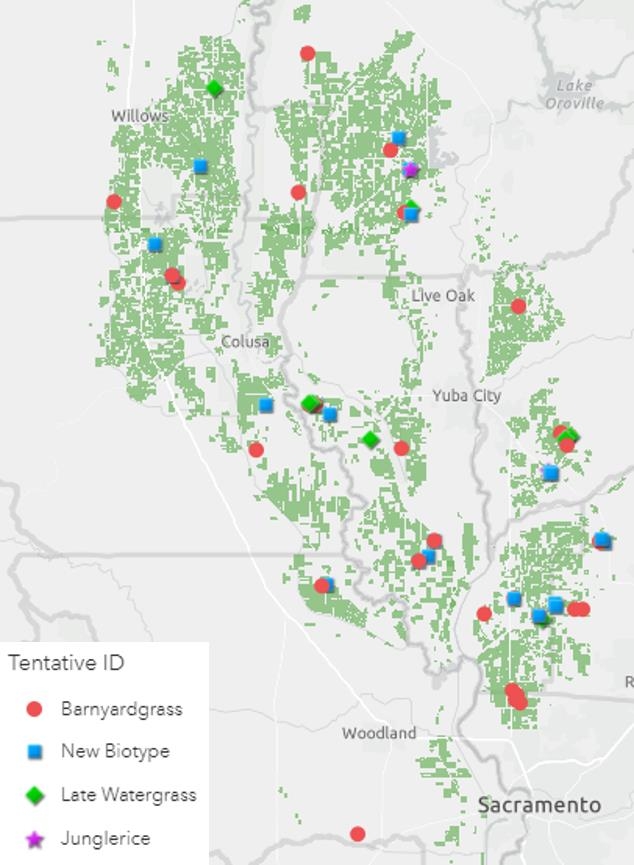
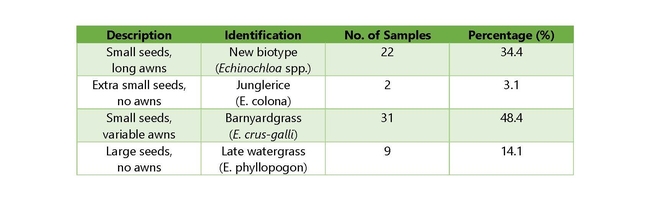
In August and September of 2020, 64 watergrass samples were collected from rice fields across the rice-growing region of California (Figure 1). The samples were representative of the Echinochloa spp. present in California rice, but were likely resistant, as they were self-reported by growers and PCAs: late watergrass (Echinochloa phyllopogon), junglerice (E. colona), barnyardgrass (E. crus-galli), and a currently unknown new biotype which is being characterized in a complementary study (Table 1). The overall objective was to determine the distribution and status of resistance to currently-registered herbicides in these species (cyhalofop, propanil, bispyribac-sodium, penoxsulam, benzobicyclon+halosulfuron, clomazone, and thiobencarb). Two known susceptible controls of late watergrass (E. phyllopogon) were added to the screenings as controls.
Screenings took place at the Rice Experiment Station greenhouse in Biggs, CA, in the summer and fall of 2021. All formulations were tested at the 1.5 leaf stage of watergrass. Dormancy was broken for the watergrass by wet-chilling in the fridge for approximately two weeks before planting. Seeds were pre-germinated in the incubator. Pots were seeded and then thinned down to 4 plants per pot.
All foliar-applied formulations (cyhalofop, propanil, and bispyribac-sodium) were applied with the label-recommended surfactants. Applications for into-the-water herbicides (granular formulations of penoxsulam, benzobicyclon+halosulfuron, clomazone, and thiobencarb) were made onto the water surface of bins that were flooded to 10 cm above the soil surface of the pots (where the watergrass was planted). All liquid herbicide treatments were applied with a cabinet track sprayer with an 8001-EVS nozzle delivering 40 gallons of spray solution per acre (at a pressure of approximately 20 psi). A flood was applied at 10 cm above the soil surface 48 hrs after the foliar applications. All herbicides were applied at standard field rates for California rice, though not at the maximum label rate for all herbicides (Table 2).
At 14 days after treatment, the number of living plants per pot was counted, and fresh biomass was measured (per pot) by cutting plants at the soil surface and taking the weight (per pot). Dry biomass was measured after drying the fresh weight samples down to a constant weight. Samples were classified as resistant to an herbicide if the average percent (%) dry weight control was less than that of the susceptible controls.
Results
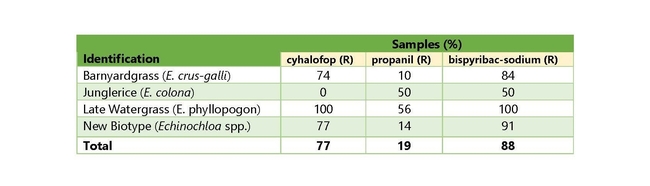
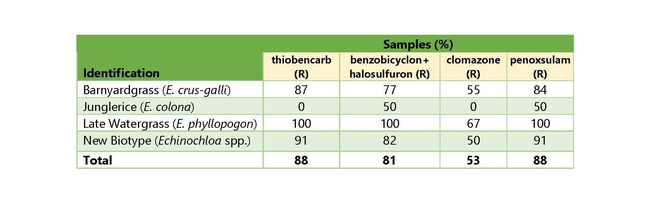
Out of the barnyardgrass samples (31), 23 were resistant to cyhalofop (CY), 3 were resistant to propanil (PR), and 26 were resistant to bispyribac-sodium (BS). Out of the late watergrass samples (9), there were 9 CY-resistant, 5 PR-resistant, and 9 BS-resistant. For the new unknown biotype samples (22), there were 17 CY-resistant, 3 PR-resistant, and 20 BS-resistant. For the granular formulations, barnyardgrass (31 samples) had 27 that were thiobencarb resistant (TH), 24 that were benzobicyclon+halosulfuron resistant (BH), 17 that were clomazone resistant (CL), and 26 that were penoxsulam resistant (PE). Out of the late watergrass samples (9), 9 were TH-resistant, 9 were BH-resistant, 6 were CL-resistant, and 9 were PE-resistant. For the new unknown biotype samples (22), there were 20 TH-resistant, 18 BH-resistant, 11 CL-resistant, and 20 PE-resistant.
The majority of the samples of all species are resistant to all of the tested herbicides, with only propanil and clomazone showing control of approximately 50% (or more) of the samples (Tables 3 and 4). Late watergrass is widely resistant to all of the herbicides tested, with only propanil showing some degree of control in roughly 50% of the samples. Surprisingly, 100% of samples tested were resistant to thiobencarb, benzobicyclon+halosulfuron, cyhalofop, bispyribac-sodium, and penoxsulam.
The new biotype is best controlled with clomazone (50% of samples) or propanil (76% of samples), while a smaller proportion of samples were controlled by the other herbicides tested. Barnyardgrass is best controlled by propanil (90% of samples), and clomazone (45% of samples).
Although the new biotype shows widespread resistance, its impact on yields is likely explained by more than just herbicide resistance and is likely due to its competitive ability as well.
Conclusions
The implications of this study reflect anecdotal evidence relayed by growers. Echinochloa spp. are becoming increasingly difficult to manage using our currently registered herbicides. For growers, this means it is increasingly difficult to plan an effective program that both controls grasses and prevents further selection for resistance. Aside from rotations with the above-utilized herbicides, some other alternative management strategies include: deep water, utilizing a stale seedbed, and rotating to a dry-seeded or drill-seeded system.
Deep Water:
Maintaining a deep flood (of at least 4–6 inches) can suppress some grass emergence. Deeper water will provide more suppression. Deep water also improves herbicide efficacy for granular herbicide applications, and the deep water may also improve efficacy of pre-emergent herbicides. Keeping the water on the field as long as possible will improve control. Watergrass typically emerges in the first 30 days after water is put on the field, so longer flood duration is better.
Stale Seedbed:
A stale seedbed has been shown to provide good control of watergrass in heavily infested fields. To implement a stale seedbed, prepare the field as normal (in spring). The field can be tilled or untilled. If untilled, please keep in mind that watergrass seeds typically only emerge from the top 6 cm (3–4 inches) of soil.
Once the seedbed is prepared, flood the field until water is 3 to 4 inches deep, then turn off water and let it sink into the soil. This will increase watergrass germination. Roughly 1 to 2 weeks later, spray a nonselective herbicide (make sure the field is fully drained to ensure coverage). Tillage can also be utilized in place of an herbicide, but avoid deep tillage, as it will bring up additional grass seeds. Timing of the herbicide application or tillage will depend on temperature. Warmer temperatures cause faster emergence of grass. Two weeks should be more than enough time to bring up most of the grass population that will be germinable (able-to-germinate), regardless of temperature.
If not planting rice, this process (flushing/flooding, followed by tillage or herbicide application) can be repeated multiple times throughout the season. If planting rice, flood up the field after the application of the nonselective herbicide (follow label for instructions on flood timing).
Rotation to Drill- or Dry-Seeded System:
Drill-seeding or dry-seeding rice allows for the use of pendimethalin, which is a different mode of action from all other currently-registered rice herbicides. Depending on the actual product used, pendimethalin may be best used in a drill-seeded system, due to the possible injury to emerging rice plants. Or it can be used in a dry-seeded system, where seed is flown on instead of drilled. For more information on application methodology, refer to the product herbicide label.
Original source: CAPCA Adviser magazine :: June 2022
- Author: Kassim Al-Khatib
- Posted by: Gale Perez
From the Rice Notes April 2022 newsletter
Kassim Al-Khatib is the Melvin D. Androus Endowed Professor for Weed Science at UC Davis and a Cooperative Extension Weed Science Specialist.
Herbicide resistance is a serious problem in California rice. However, not every control failure can be attributed to herbicide resistance. Other factors can be the cause of control failures. Among the most common reason for failure include weather, incorrect rate, poor coverage or application timing, skips, and spray equipment malfunction.
When weed control fails, it is important to determine the cause and when the cause is herbicide resistance, herbicide programs need to be adjusted. Resistance occurs after the same herbicides have been used repeatedly at the same site for several years. You will notice a gradual decline in the efficacy of the herbicide to control weeds that were once susceptible. When herbicide resistance is the problem, you will find healthy plants alongside dead ones of the same species after treatment; surviving weeds form discrete patches that consistently survive the herbicide treatment.

- Don't wait until harvest to collect the seed. By then, most weeds have shattered their seeds. If you collect after harvest, you may collect seeds from weeds that have emerged late and thus have not been exposed to the herbicide. The objective is to collect seed from plants that have survived the herbicide action.
- Collect seeds when they are mature and dislodge easily from the seedhead. In general, sprangle top matures the earliest, between rice panicle initiation and heading. Early watergrass, barnyard grass, small flower umbrellas edge, and rice field bulrush usually follow, maturing sometime before rice heading until maturity. Late watergrass matures last, at about the same time early rice varieties (M-205, M-206) mature.
- Collect seeds, not seedheads. Gently shake the seedhead inside a paper bag. Seeds that shatter are mature and will readily germinate. If seedheads are collected, seeds might not be mature or might have shattered already. It is good practice to keep the paper bag open for couple days to allow further seed drying.
- Collect seeds from areas of the field where you are certain the herbicide application in question was appropriate. Avoid field borders, tractor tire tracks, skips or areas where you suspect the herbicide was not sprayed correctly or not sprayed at all.
- Make sure to collect enough seed. In order to have conclusive results, several replications of herbicide resistance testing are needed. When not enough seed is provided, replications may not be possible. For small sized seed weed species such as sprangle top, small flower umbrellas edge or rice field bulrush, collect seeds from at least 20 mature seedheads at each location. For barnyard grass, early and late watergrass, collect from at least 30 mature seedheads.
- We will test every weed against all herbicides labeled to control that weed and you will be receiving a detailed report before the rice season start.
Original source: Rice Notes April 2022 newsletter